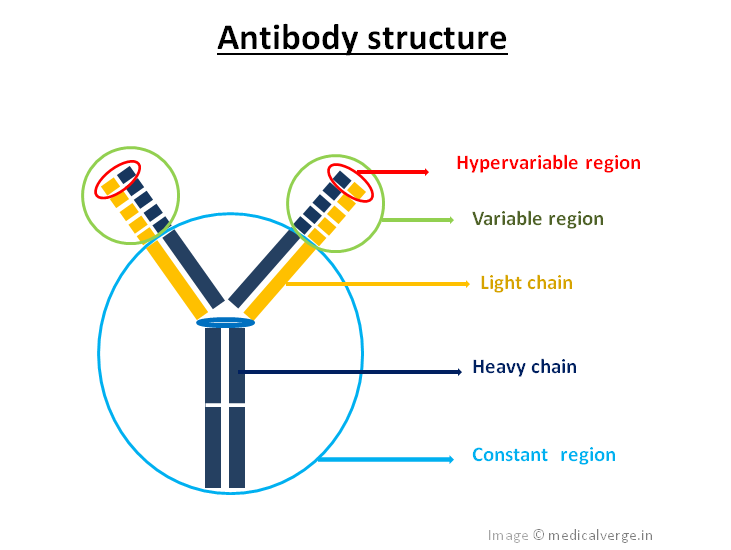
Antibodies, also known as immunoglobulins (Ig), are a type of glycoprotein formed by B-lymphocytes that can be found in a soluble form (in the blood) or membrane-bound form (on the B-cell surface). It is a major component of the humoral immune system. Antigen antibody detection mechanism is similar to a lock and key function. Each antibody has a paratope, which is a specific binding site (i.e. Lock) that binds to a specific antigen epitope (i.e. Key). A paratope is a cleft-like structure, expressed by an antibody’s hypervariable region, but also defined by the variable region. One type of B-cells produce one type of antibodies. Antibodies have a Y-shaped structure and are made up of heavy chain and light chain. The heavy chain has 3 constant (Fc) and 1 variable (VH) regions, while the light chain has only 1 constant and 1 variable (VL) region. Antibodies are classified into 5 types, based on their heavy chain. These are IgA, IgM, IgD, IgG and IgE.
Polyclonal antibodies (pAbs)
It is a set of immunoglobulins secreted by various B-cell lineages that respond to multiple epitopes of a single antigen. A prime-boost technique is used to produce pAbs by injecting an immunogen into an animal. Multiple epitope binding properties make pAbs an appealing reagent to use in research and therapeutics. They can be used directly or in refined form (through affinity column chromatography to eliminate other serum protein components). Polyclonal serum has been used to treat toxin-mediated bacterial and viral diseases for decades. Amil Adolf von Behring was awarded the Nobel Prize in 1901 for his work on serum therapy against diphtheria, which paved the way for novel treatments in medical science. Animal serum-derived therapy has been successfully used for a variety of medical conditions, including drug overdosing, viral infections (such as Rabies), and as antivenom in snake bite.
pAbs have a number of drawbacks, including limited supply, batch to batch variability, the possibility of transmitting blood-borne diseases, the fact that only a small percentage of antibodies from a set of antibodies bind to the target to exert the desired effect, the need for large doses to produce desired beneficial clinical effects, and the fact that they cannot be used to treat chronic diseases. Apart from that, polyclonal antibodies have the potential to cause allergy.
Monoclonal antibodies (mAbs)
It is generated by identical B-cells that have a high affinity and specificity for a single antigen epitope. Kohler and Milstein introduced the hybridoma technique to produce mobs in 1975 (got the Nobel prize in 1984). mAbs have very little and permissible batch to batch variation, homogeneity, and can be manufactured indefinitely. Several mAb generation technologies, such as recombinant production, bacteriophage display method, single B-cell amplification and culturing methods were later developed over time.
mAbs have played a significant role in the discovery of new targets in diagnostics and therapeutics in recent years. mAbs’ therapeutic applications are no longer limited to cancer chemotherapy and immunotherapy; a wide range of mAb-based drugs have been approved for a number of diseases, with so many more in the stage of development.
Uses of monoclonal antibodies (mAbs)
Diagnostic uses
- To detect allergies and viral diseases
- Identification of certain types of cancer as CD-marker
- Antibody based high specific imaging such as PET (Positron emission tomography, MRI (Magnetic resonance imaging), FMT (Fluorescent molecular tomography).
Therapeutic uses
- As targeted drug delivery in oncology and immunology.
- Used to produce Immunotoxins (e.g. denileukin diftitox)
- Treatment against certain viral disease (e.g. HIV, RSV)
- As a wide variety of cancer chemotherapy (e.g. breast cancer, lymphomas)
- Treatment of various autoimmune disorders (e.g. multiple sclerosis)
Research field
- Used in labeling to identify some specialized cells, such as neuron.
- Used in purification of complex biological mixture and structural cell membrane protein.