The scientific study of the effects of time on living systems and biological rhythms is known as chronobiology. Chronopharmacology is a branch of Chronobiology concerned with the effect of drugs upon timing and rhythm of biological events in order to enhance therapeutic efficacy while minimizing side effects.
Circadian Rhythm
Circadian is a Latin word, circa means ‘about’ and dian means ‘day or about 24 hours’. The word ‘circadian’ was coined by Franz Halberg in the 1960s. Circadian rhythm is defined as “oscillation in the biological, physiological and behavioral function of an organism with a periodicity of 24 hours”. In the 18th century, Jean-Jacques d’ortous de mairan was the first to describe the circadian rhythm in plants. In 1918, J.S. Szymanski described the circadian rhythm in animals.
Many scientists believe that melatonin peaks and troughs over time play an integral role in synchronizing the body’s circadian rhythm with the external cycle of light and darkness. Environmental cues (e.g., light-darkness, warm-cold, noise-quiet, social contact-isolation) emerge in tandem with the sleep-wake cycle, and are referred to as synchronizers or zeitgebers.
Photoreceptor cells in the retina perceive light, which is then conveyed to the suprachiasmatic nuclei (SCN) via the retinohypothalamic tract. The molecular clockworks rely heavily on clock genes. The mechanism by which circadian output pathways work is unknown, though it is thought to entail both neurological and hormonal signals. Jeffrey C. Hall, Michael Rosbash, and Michael W. Young Won were awarded the Nobel Prize in 2017 for isolating a gene that helps in managing the body’s clock in fruit flies, which have a genetic composition remarkably similar to humans. This research suggests that “Cryptochrome genes,” which code for proteins that accumulate at night and degrade during the day, are accountable for the sensation of being awake
Factors influencing circadian rhythm
- A mutation in one or more genes
- Jet lag or shift work
- Light form electric gadgets at night
These variables can interfere with our biological clock, but it will eventually adjust to the new time zone. Traveling (flying) from east to west results in the loss of hours, but travelling (flying) from west to east results in the gain of hours. According to a study, adjusting for time gain is easier than adjusting for time loss.
Biological rhythm observed in various biological systems
| Peak level | Timing rhythm |
|
Serum Cortisol, Aldosterone, Testosterone, Platelet adhesiveness, Blood viscosity, NK-cells activity |
Peak level during the initial hours of daytime. [Morning] |
| Haematocrit (RBC, Hb) | Highest around middle of daytime. [Noon] |
| Airway caliber (FEV1) | Greatest around middle of daytime. [Afternoon] |
| Insulin, Cholesterol, Triglyceride, Platelet number, Uric acid | Peak during the late hours of day. [Evening] |
| Basal gastric acid secretion, WBC (lymphocyte, eosinophil), Prolactin, Melatonin, ACTH, LH, FSH | Peak level at specific times during the night. [Night] |
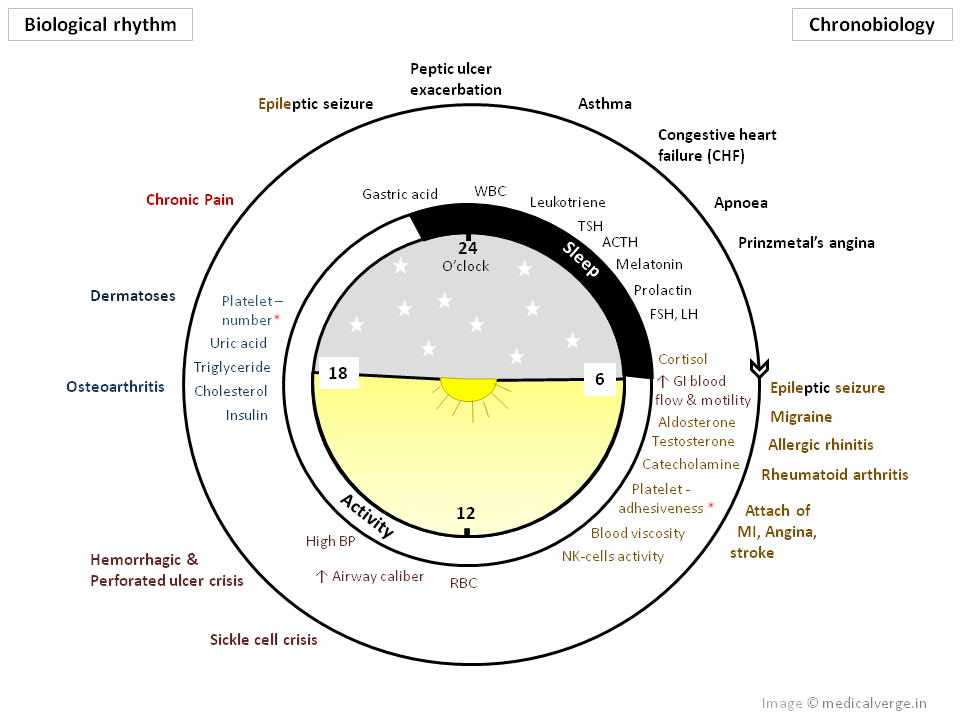
biological rhythm in disease occurrence
| Diseases | Rhythm on exacerbation |
| Allergic rhinitis | Worst in the morning upon awakening |
| Migraine headache | Provoke most frequent in the morning at the time of awakening |
|
Thromboembolic/ischemic events like MI, Angina Ischemic/ hemorrhagic events of Stroke |
Highest during the initial hours of daytime, around the time of commencing daily activity |
| Rheumatoid arthritis | Symptoms worse in the morning |
| Osteoarthritis | Worst later in the day around evening |
| Hypertension | BP is highest in the afternoon, which gradually decreases in the morning, and attains lowest value at night. |
| Congestive heart failure (CHF) | Worse nocturnally |
| Epilepsy | Epileptic seizure is more common around the time of sleep at night and in the early morning. |
Chronopharmacokinetics
It deals with the study of the temporal changes in absorption, distribution, metabolism and elimination of drugs. This takes into account the impact of drug administration time on these different steps: –
Absorption
The absorption of various drugs, notably lipophilic drugs, is affected by diurnal variations in gastric acid secretion (pH), gastric motility, and gastrointestinal blood flow. Lipophilic drugs are absorbed faster in the morning.
Distribution
A putative difference in drug distribution could be explained by circadian variations in plasma protein binding and blood flow. Circadian variation in sympathetic and parasympathetic system activity may be a pivotal regulatory mechanism for diurnal increase and nocturnal decrease in blood flow.
Metabolism
There is circadian variation in enzyme activity and hepatic blood flow. The liver is a biological clock capable of generating its own circadian rhythm. Clock gene plays a key role in the molecular clockworks of both the SCN and liver. The liver is a major organ for metabolism and detoxification, hence coordinated rhythmic oscillation in both phase I and phase II components may explain the differential responses to drug metabolism.
Hepatic enzymes
There is a considerable circadian rhythm observed in drug metabolizing enzymes: –
Phase-I metabolism – Cytochrome P-450 (CYPP17, CYP2A4, CYP2E1, CYP2C2)
Phase-II metabolism – N-acetyl transferase Camello 4 (Clm 4), Glutathione S-transferase, Carboxylesterse
Ion transporter genes
The expression of different genes encoding proteins involved in ion transport obeys a circadian rhythm. Some examples are: –
– Solute carrier/sodium potassium chloride cotransporter (Slc) genes such as Slc12a2, Slc16a1, Slc19a1 etc.
– Potassium channel transporter genes like HCN-4
– Sodium channel transporter genes like Scn4a, Scn2b
– Copper transporter genes like Atp7b
Excretion
Circadian variation in renal excretion may be due variation in GFR, renal blood flow, urinary pH and tubular resorption. Urinary pH influence drug ionization, hence its excretion. Acidic drugs such as sodium salicylate and sulfasalazine are excreted faster after an evening administration.
Circadian variability in pharmacokinetics of drugs
Antiasthmatic
Dosing theophylline in the morning (8 o’clock/8 AM) produces higher peak plasma concentrations than dosing in the evening (20 o’clock/8 PM). As a result, the amount necessary in the evening is higher than during the day, when asthma is primarily nocturnal. H1 antihistaminics such as cyperoheptadine, clemastine, and terfenadine exert longer duration of action in the morning (7 AM) than in the evening (7 AM). Leukotriene modulators such as montelukast, zafirlukast, and zileuton are most effective in a night dose, because leukotriene concentration is highest at night.
Antibiotic
Amikacin attains a greater serum level Around 9 PM. Gentamicin shows long half-life between 8 PM to midnight. Sulfamethoxazole shows longer half-life at midnight. Renal excretion of ciprofloxacin is lower at night around 10 PM.
Cancer cells are more susceptible to S-phase specific agents like (5-fluorouracil) at night, while normally rapidly dividing cells in the body, such as skin and bone marrow, are less susceptible by 50% between 12 PM to 4 AM, indicating that night dosage is more tolerable. Cisplatin, a DNA-binding agent, has a lower renal toxicity and is better tolerated in the evening (4PM to 8 PM).
Antiulcer
H2 blockers such as ranitidine are given once a day in the evening or night, when basal gastric acid secretion is high. In contrast to PPIs like pantaprazole, rabeprazole has a better regulation of gastric pH in the morning than in the evening.
Antihypertensive
After morning dosage, cardiovascular drugs such as nifedipine, propranalol, and oral nitrate achieve higher plasma peak concentrations (Cmax and Tmax). This could be owing to the fact that stomach emptying is faster and GI perfusion is higher in the morning. On morning dosing (before (9 AM), thiazides had a decreased risk of hypokalemia.
Opioid analgesic
Opioids, like tramadol and dihydrocodeine have a greater analgesic effect when administered in the evening.
Local anesthetic
Amide type local anesthetics (e.g. lidocaine, ropivacaine, and mepivacaine) have the longest duration of action, when employed in the afternoon (around 3 PM). Lidocaine attains higher plasma concentration in the evening. Bupivacaine has high clearance at early morning (6 AM).
General anesthetic
Benzodiazepines like midazolam shows shortest elimination half-life when given at afternoon (2 PM). When barbiturates like pentobarbital were administered in mice during the dark phase, the plasma concentration was significantly greater as compared to light phase.
Chronopharmacodynamics
Biological rhythm determinants at the cellular and subcellular levels can cause variations in drug efficacy and vulnerability to side effects. The diurnal variations in drug pharmacodynamics could be explained by rhythms in receptor number or its structural conformation, second messengers, metabolic pathways, and plasma protein binding. This phenomenon is more important in toxicology (Chronotoxicity). Examples: –
- Prednisolone/methylprednisolone – the morning dose of Prednisolone has a decreased risk of adrenocortical suppression, when compared to any other dosage time.
- Irinotecan hydrochloride – adverse effects such as leukopenia and body weight loss were more severe in the active phase than in the resting phase (in mice).
- IFN-β – the anticancer effect of IFN-β was more pronounced during the early resting phase than during the early active phase.
- IFN-α – has shown a greater antiviral and lymphocyte-stimulating effect during the early resting phase than during the early active phase.
Chronotherapeutics
It is a field in which drug administration is synchronized with the biological rhythm in order to optimize the therapeutic effect while minimizing side effects. A collective knowledge of variation in disease intensity and exacerbation, impact of circadian rhythm on the efficacy and safety of medication constitutes the rationale for a novel pharmacological approach.

Examples of chronotherapeutics in the medical practice
- Corticosteroids are administered either in the morning (8 a.m.) or in the afternoon (around 3 PM).
- Theophylline and Leukotriene modulators are given in the evening for nocturnal asthma.
- H2-blockers are taken once a day in the evening or night, while PPIs are taken once a day in the morning, half an hour before a meal, on an empty stomach.
- Aspirin is administered at bedtime, because platelet adhesiveness in higher in the early morning.
- Administration of Thiazide diuretics in the morning (before 9 AM) has a lower risk of hypokalemia than any other dosing time.
- Using testosterone medication delivery patches in the morning to accomplish physiological androgen replacement.
- Hypnotics (e.g. clonazepam, zolpidem) and melatonin agonist (like ramelteon) are given before bedtime for insomnia.
- Calcium channel blockers (e.g. nicardipine, amlodipine) preferably administered in the evening, when used for CCF.